My asthma is not manageable
Monitor and Alert
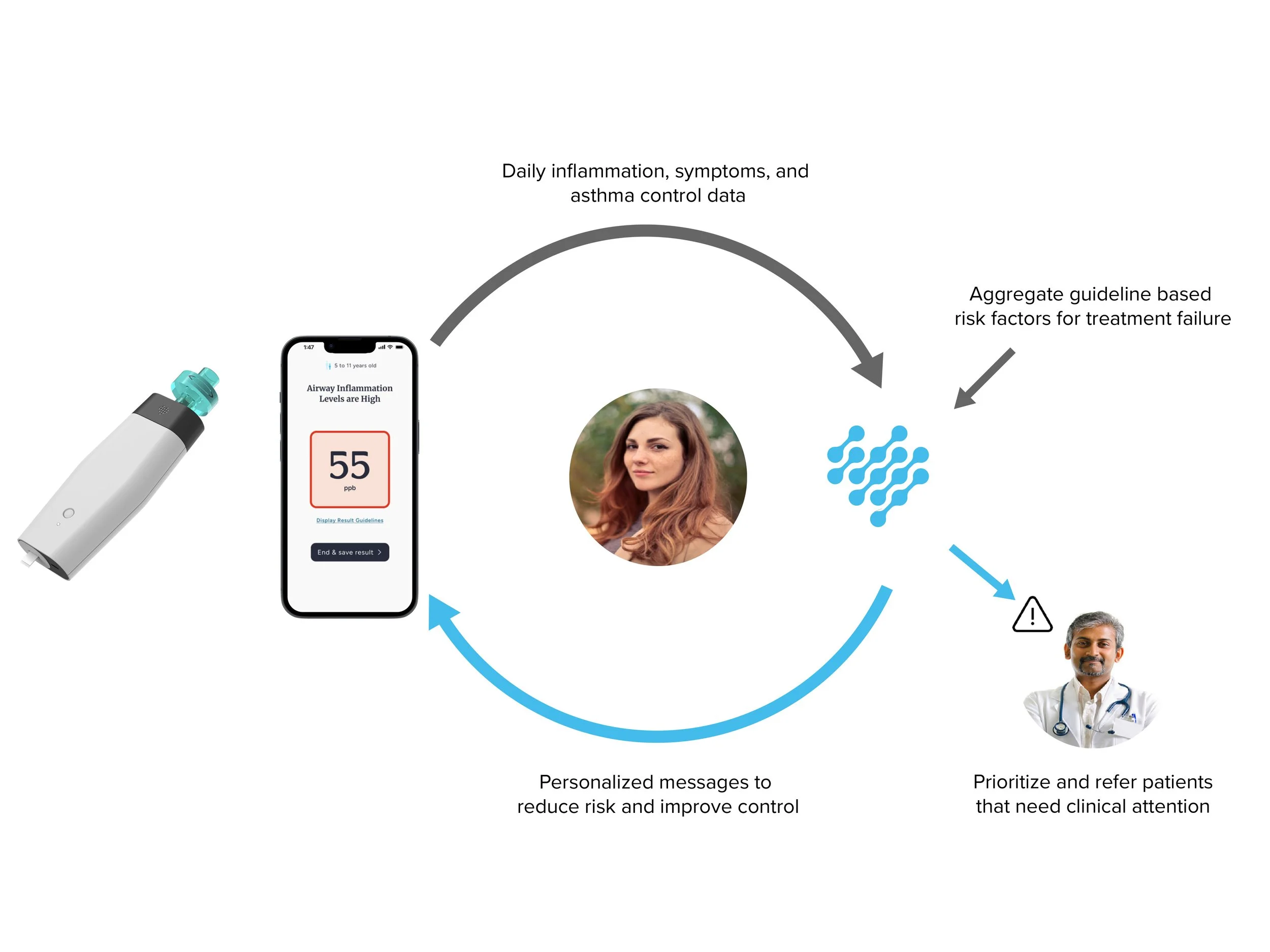
Our app gathers clinical and environmental risk factors to determine the root cause of symptoms and poor asthma control.
myBiometry Prediction Engine
We aggregate all the guideline-based risk factors associated with uncontrolled symptoms and attacks to build personalized, predictive risk models for each patient.
Actionable Insights
We put each result into context so patients can take action to lower their risk of attack and improve asthma control.
We leverage our own data to proactively engage patients before symptoms and attacks occur to deliver guideline-based coaching and education to improve self-management.
Education at Scale
Guideline based self-management programs delivered in app and scalable to millions of patients. Programs are recognized by the CDC to improve control and reduce costs.